Outsourced Manufacturing for Oral Dosage Forms
From the commonly available tablets and capsules to more specialized forms like bi-layer and chewables, oral dosage forms remain the most popular drug products on the market. In this highly competitive and flourishing landscape, biopharma companies and their service providers rely on the most current information to differentiate themselves, proactively assess risks, and align their day-to-day operations with their strategic goals.
Industry Standard Research (ISR Reports) has studied the market dynamics of outsourced oral dosage forms manufacturing since 2015, facilitating informed business decisions for drug innovators and contact manufacturers (CMOs), as well as their financial sponsors and consultants. Our latest research revealed our biopharma respondents’ desire for the latest oral dose technologies and the key provider capabilities they seek when planning for the success of their outsourced oral dose manufacturing projects.
Oral Dose Technologies Increasing in Demand
The fastest growing oral dose technologies are all emerging or niche technologies. Most impressively, utilization of Plant-based polymers is expected to nearly triple from 9% today to 26% in the next five years. Industry concern with environmentally sustainable manufacturing indicates a shift toward plant-derived bio-based polymeric materials (or “next-generation polymers”)1, aligning with what participants revealed in our survey.
One-third of respondents currently utilize Formulation solutions for poorly soluble compounds. By 2028, nearly one-half of respondents expect their company to use these technologies, a growth of 15 percentage points. Utilization of Enteric or targeted release technologies and Combination therapies are projected to increase by 12 and 9 percentage points, respectively.
“Select each of the technologies your company currently utilizes for oral dosage drug products, whether those products are marketed or in development. Select all that apply.” (n=104)
“Five years from now, which technologies do you think your company will utilize for oral dosage drug products that are marketed or in development? Select all that apply.” (n=104)
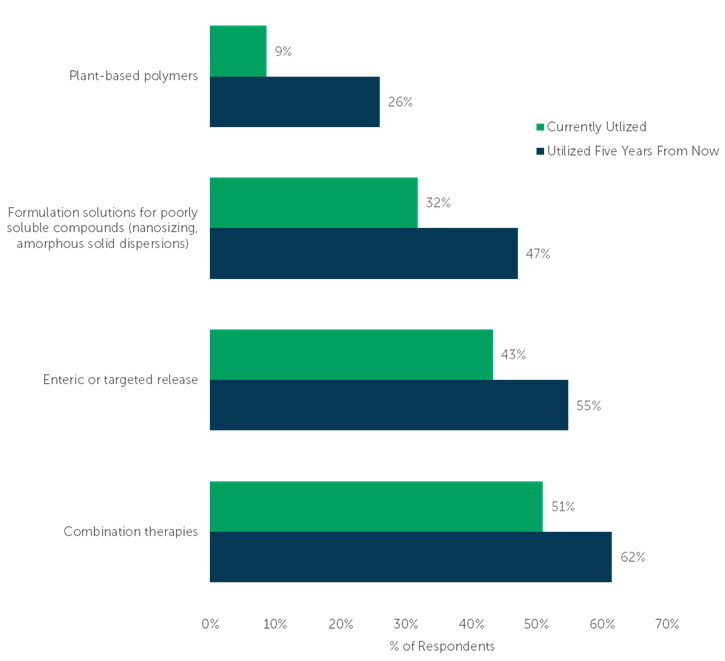
Chart sorted by magnitude of projected growth.
Service Provider Capabilities Increasing in Demand
It’s critical for biopharma companies to select manufacturers that can adequately and efficiently meet the needs of their oral dose projects. CMOs marketing themselves to drug innovators would do well to demonstrate their proficiency around the key capabilities that sponsors seek in a service provider to win and keep their business.
Top spray granulation tops the list of service provider capabilities that will increase in demand over the next five years. 22% of respondents currently require Top spray granulation from their CMO; 36% anticipate needing this capability from their service provider five years from now. Demand for Direct pelletization, High-sheer granulation, and Roller compaction are expected to grow 12 percentage points each. Oven tray drying / curing rounds out the top-five in-demand service provider capabilities, increasing by 11 percentage points.
“Which of the following capabilities does your company currently require when engaging a contract manufacturer for an oral dosage manufacturing project? Select all that apply.” (n=104)
“Five years from now, which of the following capabilities do you anticipate your company requiring when engaging a CMO for an oral dosage manufacturing project? Select all that apply.” (n=104)
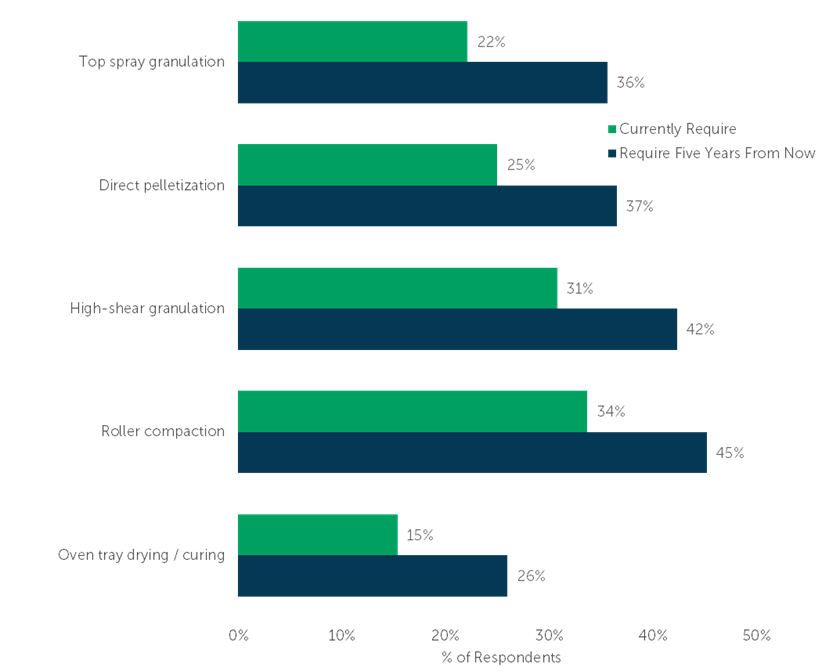
Chart sorted by magnitude of projected growth.
Oral Dosage Forms Market Outlook
Our research shares insights from outsourcers on what works and what doesn’t with regard to their oral drug product manufacturing endeavors. Reliable and actionable market research provides a data-driven foundation for drug innovators to forge beneficial partnerships and enables service providers to better understand their customers so they can do the same.
The full Oral Dosage Forms Market Outlook (5th Ed.) report includes key statistics such as oral dose project volume and percentage outsourced, the average number of CMOs needed to complete meet their oral dose manufacturing needs, and a detailed analysis of 17 different oral dosage forms. Armed with novel findings that are backed by ISR’s rigorous survey methodologies, professionals that navigate the oral dose manufacturing space can act with confidence and make the most of their outsourced drug development activities.
Primary market research data in this article were powered by the ISR Health Panel. Want to contribute to thought leadership pieces and help to make the pharma industry better? Join today.
Learn More Oral Dosage Forms Market Outlook (5th Ed.)
1Joseph, T.M.; Unni, A.B.; Joshy, K.S.; Kar Mahapatra, D.; Haponiuk, J.; Thomas, S. Emerging Bio-Based Polymers from Lab to Market: Current Strategies, Market Dynamics and Research Trends. C 2023, 9, 30. https://doi.org/10.3390/c9010030





